
Medicines optimisation is a person centred way of choosing, using, reviewing and, when needed, stopping medicines so people get the best possible benefit with the least avoidable harm.
It goes further than prescribing. A medicine may suit a condition and still fail in day to day life. The timing may be unrealistic. Side effects may become too much. The person may not see the point of taking it. The dose may no longer suit their health, age or frailty.
That is why medicines optimisation sits at the centre of good care. It brings together evidence, safety, review and shared decisions. It asks whether treatment still helps, whether it still fits, and whether the person is getting real benefit from it.
This approach runs through NHS England’s medicines optimisation work, NICE guidance on the safe and effective use of medicines, NICE guidance on medicines adherence, NICE guidance on multimorbidity, and the MHRA Yellow Card scheme. It also connects with the Human Medicines Regulations 2012, the Mental Capacity Act 2005, the Equality Act 2010 and the Health and Social Care Act 2008 (Regulated Activities) Regulations 2014.
“A medicine only earns its place when it still helps the person taking it.”
For health and social care services, this means fewer avoidable problems, better symptom control and less waste. For people receiving care, it can mean a simpler routine, fewer side effects and more confidence in treatment. Shorter lists. Better use.
Why Long Term Conditions and Polypharmacy Have Raised the Stakes
Medicines optimisation has become more prominent because more people now live for years with several long term conditions. A person may have diabetes, heart failure, arthritis, chronic lung disease and depression at the same time. Each condition may bring its own medicines, monitoring and follow up.
That creates a heavy treatment load. It is not unusual for one person to manage tablets, inhalers, eye drops, creams and occasional medicines, all with different timings and instructions. The list can be clinically correct and still feel unmanageable.
Polypharmacy means taking multiple medicines. This is not always a problem. Plenty of people need several medicines and do well on them. Trouble starts when the whole regimen becomes too complex, too risky or too burdensome to manage well.
Put more plainly, the issue is not just the number of medicines. It is the overall fit between the medicines, the person’s health and the life they are trying to live.
A resident in a care home may take regular tablets for blood pressure, heart disease, diabetes and pain, plus a sleeping tablet and a laxative. Staff may then notice drowsiness, constipation and unsteadiness. A review may show that one medicine is no longer helping, another is adding to falls risk, and the sleep medicine has stayed on repeat without a fresh look.
Common terms in this area include:
- Multimorbidity: Two or more long term conditions in the same person.
- Polypharmacy: Several medicines taken at the same time.
- Treatment burden: The work involved in managing treatment, reviews and monitoring.
- Appropriate polypharmacy: A medicine regimen with a clear overall benefit.
- Problematic polypharmacy: A regimen where the burden or risk starts to outweigh the benefit.
There is a fair point to make here. Several medicines can be the right answer, especially in cardiovascular disease, diabetes or severe mental illness. Medicines optimisation is not about cutting numbers for the sake of it. It is about having a clear reason for each medicine and checking that reason still holds.
What Non Adherence Looks Like and Why It Leads to Waste
Non adherence means the person is not taking medicines in the way that has been agreed. This can involve missed doses, extra doses, stopping early, taking medicine at the wrong time, or using a device badly.
The reasons are often ordinary. A person may forget the lunchtime dose. They may stop after reading a leaflet that worries them. They may struggle to open the packet. They may not believe the medicine is doing much. They may feel worse after taking it and keep quiet about it.
The result is often the same. Poor symptom control. Treatment failure. More appointments. Wasted medicines.
“Unused medicines often point to a treatment plan that never really fitted.”
These patterns turn up across settings:
- At home: A person misses evening tablets because they go to bed early and no longer follow the routine they once had.
- In supported living: Weekend doses are often missed because weekday prompts are stronger than weekend support.
- In a care home: A resident refuses a capsule because it is large and unpleasant to swallow.
- After discharge: The old medicine and the new medicine are both taken because the change was not explained clearly.
Waste is not only about cost. It reflects missed benefit and missed chances to improve care. Cupboards full of unused medicines, repeated requests for medicines that are no longer taken, or boxes returned unopened all point to the same problem. The plan did not land well.
A worker might put it like this: “She’ll take it if we sit with her and explain it, but not if it is left on the table and rushed through”. That sort of comment tells you more than a stock count ever will.
A sensible response is usually simple and structured:
- Spot the pattern: Look for late repeat requests, stockpiled boxes, frequent refusal or worsening symptoms.
- Ask without blame: A calm conversation usually reveals more than a warning ever will.
- Find the barrier: Side effects, poor memory, packaging, routine and fear are all common causes.
- Adjust the plan: A review, a simpler schedule, a different formulation or better explanation may help.
- Check again: A change only counts if it actually improves use and outcomes.
This is where NICE guidance on adherence, the Care Certificate standards and day to day professional judgement come together. A prescription on a screen does not show whether the person can live with it.
How the Main Aims of Medicines Optimisation Fit Together
The aims of medicines optimisation are closely linked. Better communication can improve adherence, reduce waste and make treatment safer. A well timed review can remove a medicine that no longer helps, which may then reduce side effects and simplify the whole regimen.
The main aims are usually described in practical terms:
- Improve outcomes: Treatment should help the person in a meaningful way.
- Support informed use: The person should know what the medicine is for and what to expect.
- Reduce avoidable harm: Side effects, interactions and errors should be spotted early.
- Avoid unnecessary treatment: Medicines that do not help or duplicate others should be reviewed.
- Reduce waste: Supply and use should reflect real need.
- Promote value: Medicines should offer sound clinical benefit for the resources used.
Take a familiar example from primary care. A GP practice reviews a person taking several repeat pain medicines. One no longer helps. One causes daytime drowsiness. One was started after a short term flare up and never stopped. The person also buys over the counter pain relief. A good review can cut duplication, reduce sedation and make the routine easier. It may also lower the chance of falls or constipation.
The same logic applies in a hospital ward, a care home, a community clinic or a person’s own home. Medicines optimisation is not an extra layer placed on top of care. It is part of careful care.
A useful checklist includes:
- Purpose: Does each medicine still have a clear reason for being there?
- Benefit: Is it helping enough to justify the burden?
- Safety: Are there side effects, interactions or monitoring gaps?
- Fit: Can the person manage the medicine as intended?
- Review: Has anything changed since the medicine was started?
“The best medicine plan is not the longest one. It is the one that still fits.”
How Shared Decision Making Shapes Better Treatment
Shared decision making sits at the heart of medicines optimisation. The clinician brings evidence, options and judgement. The person brings lived experience, preferences and priorities. Good treatment plans need both.
People do not all want the same thing from care. One person may accept side effects to gain longer term protection against stroke or heart attack. Another may place more value on staying alert, mobile or independent each day. Those differences shape the right choice.
This becomes especially important in long term conditions and frailty. A medicine that lowers risk over years may not be the best fit for someone whose treatment burden is already high or whose goals have changed. That does not make the medicine wrong in general. It means the decision must be specific to the person.
Shared decision making works best when it includes:
- Goals: What does the person want treatment to achieve?
- Options: What are the realistic choices, including doing less or stopping something?
- Benefits and risks: What is likely to improve, and what may go wrong?
- Practicalities: Can the medicine be taken, used or monitored properly?
- Review points: When will the effect be checked again?
A carer might describe the difference very simply: “Once someone had taken the time to explain why it was there, he stopped saying no to it every morning”. That is not a dramatic story. It is ordinary care done properly.
This also links with legal and professional duties. The Mental Capacity Act 2005 applies where capacity is in question for a specific decision. The Equality Act 2010 is relevant where reasonable adjustments or accessible information are needed. The Care Act 2014 frames wider duties around wellbeing and person centred care, while SCIE guidance on mental capacity supports good practice in care settings. The General Medical Council’s guidance on prescribing and managing medicines also supports safe and person centred prescribing.
A realistic example is anticoagulation for atrial fibrillation. The discussion needs more than a brief statement that the medicine reduces stroke risk. It should also cover bleeding risk, monitoring needs where relevant, missed doses, daily routine and the person’s own priorities. That is what makes the decision meaningful.
What Evidence Based Choice and Cost Effectiveness Look Like
Medicines optimisation is person centred, but it is not based on preference alone. Medicines still need to be chosen on sound evidence. That means looking at effectiveness, safety and cost effectiveness together.
Cost effectiveness can sound dry, yet the idea is simple. A medicine should offer worthwhile benefit for the resources it uses. Cheap treatment is not always good value if it causes side effects, poor adherence or extra appointments. A slightly more expensive option may sometimes be better value if it works better or is easier to use.
A few examples bring this to life:
- Generic prescribing: A suitable generic medicine may provide the same benefit at lower cost.
- Reviewing duplication: Two medicines from the same group may raise risk without adding benefit.
- Choosing the right formulation: A liquid, patch or dispersible tablet may suit someone with swallowing problems.
- Stopping low value treatment: A medicine that no longer helps may add burden without real gain.
- Better antibiotic use: Careful prescribing supports antimicrobial stewardship and reduces unnecessary exposure.
This section often gets reduced to price alone. It is broader than that. Cost effective does not mean choosing the cheapest option regardless of the person in front of you. It means choosing treatment that is clinically appropriate and sensible in the round.
A surgery reviewing repeat antibiotics, sedatives or long term pain medicines may see this clearly. The issue is not only volume. It is whether the medicine is still indicated, whether the duration is right, and whether there is a better alternative. The same applies to branded medicines where suitable generic options exist, and to short term medicines that have drifted into long term use.
Useful reference points include NHS England’s medicines optimisation pages, NICE guidance on medicines optimisation and the BNF’s medicines guidance.
How Safer Medicines Use Is Built into Everyday Care
Safety is one of the clearest parts of medicines optimisation. Medicines can help, but they can also cause harm through side effects, interactions, allergy problems, dose errors, missed monitoring and confusion during transfers of care.
Safe systems reduce those risks. They rely on careful records, clear communication and regular review. Nothing flashy. Just consistent good work.
A few processes stand out:
- Medicines reconciliation: The full list is checked when a person is admitted, discharged or transferred.
- Structured review: The current regimen is examined for benefit, safety, burden and ongoing need.
- Clear records: Dose changes, stopped medicines and monitoring plans are recorded properly.
- Monitoring: Blood tests, observations and symptom checks happen when required.
- Incident reporting: Errors and near misses are used to improve systems.
- Side effect reporting: Staff and patients can use the Yellow Card scheme to report suspected adverse effects.
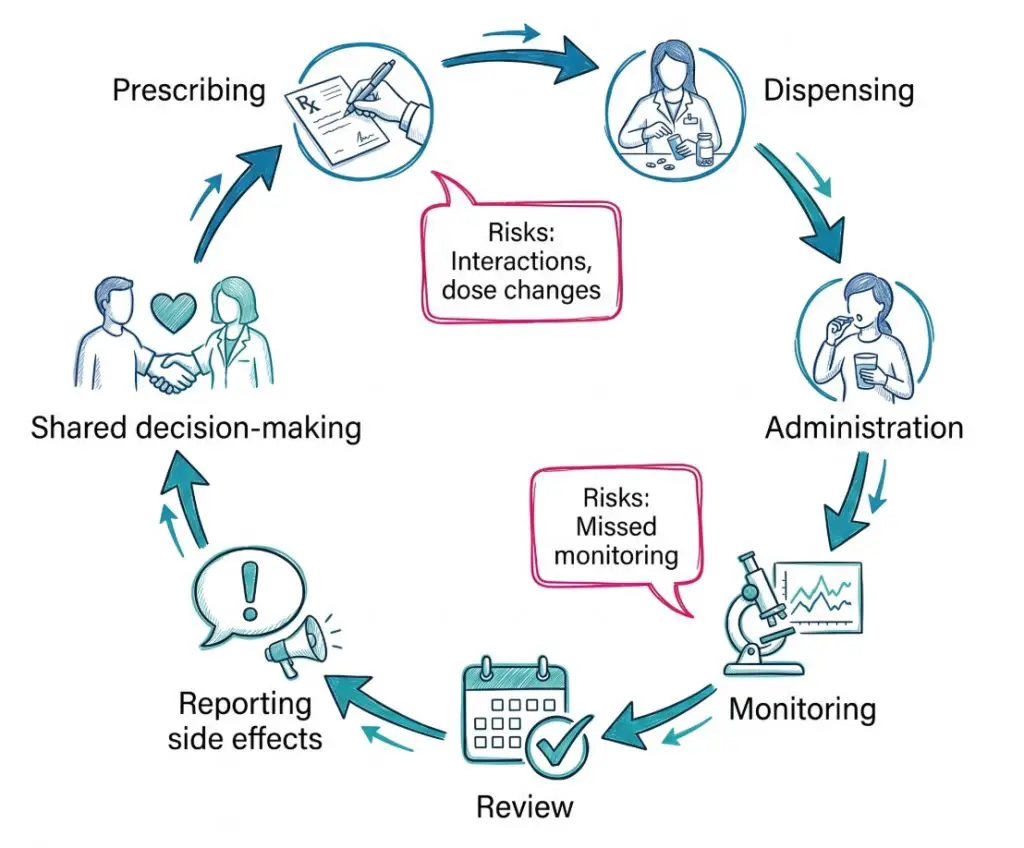
The legal and regulatory background supports this work. The Human Medicines Regulations 2012 set the wider framework for the sale, supply and administration of medicines. CQC guidance on medicines management links medicines use with safe care. Regulation 12 of the Health and Social Care Act 2008 (Regulated Activities) Regulations 2014 requires care and treatment to be provided in a safe way. The Health and Safety Executive also provides wider guidance relevant to safe systems of work in health and social care.
Picture a hospital discharge. A person leaves with a changed insulin dose and one blood pressure medicine stopped. If the GP record is not updated, the community pharmacy may dispense the old item, and family carers may continue the previous routine. One gap becomes several.
A care home example is just as familiar. Staff notice a resident is more sleepy, slower on their feet and less interested in meals after several medicine changes. Those signs may point to a medicines problem rather than ‘just age’. Timely review can prevent a fall, dehydration or admission.
“Safe medicines use depends on good systems, not good luck.”
Where Medicines Optimisation Happens Across Health and Social Care
Medicines optimisation should be built into routine care. It does not belong to one profession or one setting.
In general practice, it often appears through repeat prescribing, chronic disease reviews and structured medication reviews. In hospital, it depends on accurate admission histories, ward pharmacy input and clear discharge communication. In community pharmacy, it appears in counselling, medicine queries, interaction checks and support with self management.
Care homes and home care services bring another layer. Staff there see what happens between appointments. They see refusal, confusion, drowsiness, constipation, poor inhaler technique and difficulty opening packets. Those observations are often the first sign that treatment no longer fits well.
A brief comparison helps:
| Setting | Typical medicines optimisation activity | Common risk if missed |
|---|---|---|
| General practice | Repeat review, structured medication review, safer prescribing checks | Medicines stay on repeat without proper review |
| Hospital | Reconciliation, dose changes, discharge communication | Duplication, omission, unclear changes after discharge |
| Care home | Administration, observation, escalation of concerns | Side effects and refusal go unnoticed |
| Community pharmacy | Advice, counselling, supply checks, interaction spotting | Problems with use remain hidden until harm occurs |
| Home care or supported living | Prompting, administration support, reporting barriers | Weekend gaps, missed doses, poor routine fit |
This is one reason medicines optimisation has real depth as a topic. It is not a policy slogan. It describes a set of actions that look different in each setting but aim at the same outcome.
Who Contributes to Medicines Optimisation
Medicines optimisation works best when responsibility is shared clearly. No single professional sees the full picture.
Key roles include:
- Prescribers: GPs, nurse prescribers, pharmacist prescribers and other authorised prescribers choose, review and adjust treatment.
- Pharmacists: They support prescribing, review, counselling, reconciliation and quality improvement.
- Pharmacy technicians: They help with systems, supply, reconciliation and practical medicines processes.
- Nurses: They administer medicines, monitor effects and raise concerns.
- Care workers and support workers: They often spot the first day to day signs that a regimen is not working.
- Patients, families and carers: They live with the medicines and often notice side effects or barriers first.
Professional bodies and organisations shape the wider framework. NICE produces guidance. NHS England leads national work on medicines optimisation. CQC regulates services. MHRA oversees medicines safety. The General Pharmaceutical Council sets standards for pharmacy professionals and registered pharmacies. Skills for Care supports workforce learning in adult social care. SCIE provides practice resources that support person centred care and safe decision making.
A useful mini case study is a care home resident returning from hospital on changed medicines. The doctor writes the discharge summary. The ward pharmacist checks the list. The GP updates the record. The community pharmacy dispenses the new items. Care home staff administer and observe. If one part fails, risk rises. If each part works well, the person is far safer.
What Common Mistakes Get in the Way
The most common mistakes are ordinary ones. That is exactly why they keep happening.
Examples include:
- Assuming repeat means appropriate: A medicine can continue for years without anyone asking if it still helps.
- Looking only at the prescription: A treatment plan may be clinically sound and still impossible to manage.
- Treating refusal as poor behaviour: Refusal may reflect side effects, fear, confusion or swallowing difficulty.
- Missing transfer risks: Admission, discharge and specialist reviews can create duplication or omission.
- Ignoring formulation and technique: The right medicine may still fail if the person cannot swallow it or use the device.
- Keeping low value treatment by habit: Medicines sometimes stay because stopping them feels harder than reviewing them.
A short, practical checklist can reduce these problems:
- Check purpose: Why is each medicine still being used?
- Check benefit: Is it helping enough to justify the burden?
- Check safety: Are there side effects, interactions or monitoring needs?
- Check fit: Can the person actually take or use it as intended?
- Check recent change: Has there been a discharge, clinic change or new diagnosis?
- Check review date: When was the last proper review?
One caution is worth keeping in view. A long medicines list is not automatically poor care. There are many cases where several medicines are clearly justified. The problem is not the list itself. It is the absence of a fresh, informed reason for each item on it.
Conclusion
Medicines optimisation is about getting the best possible benefit from medicines while reducing avoidable harm, waste and unnecessary complexity. It links good prescribing with good review, safe systems and treatment that fits everyday life.
That is why the topic stays relevant across general practice, hospitals, community pharmacy, care homes and support at home. A medicine may be evidence based, licensed and properly supplied, yet still fail the person taking it. Review, communication and shared decisions close that gap.
The strongest medicines plans are usually the clearest ones. Each medicine has a reason. The person knows what it is for. Risks are watched. Changes are recorded. Treatment that no longer helps is reconsidered.
The next step for any service looking at medicines use is straightforward. Better choices, better review and better follow through.
Subscribe to Newsletter
Get the latest news and updates from Care Learning and be first to know about our free courses when they launch.
